GE OEC 9800
GE Healthcare is a subsidiary of American multinational conglomerate General Electric incorporated in
New York and headquartered in Chicago, As of 2017, the company is a manufacturer and distributor of
diagnostic imaging agents and radiopharmaceuticals for imaging modalities that are used in medical imaging procedures.
The company offers dyes that are used in magnetic-resonance-imaging procedures. GE Healthcare also manufactures medical
diagnostic equipment including CT image machines. Further, it develops Health technology for medical imaging and information
technologies, medical diagnostics, patient monitoring systems, disease research, drug discovery, and biopharmaceutical manufacturing.
The company was incorporated in 1994 and operates in more than 100 countries. GE Healthcare operates as a subsidiary of General Electric.
NEW PARTS SYSTEN IN STOCK
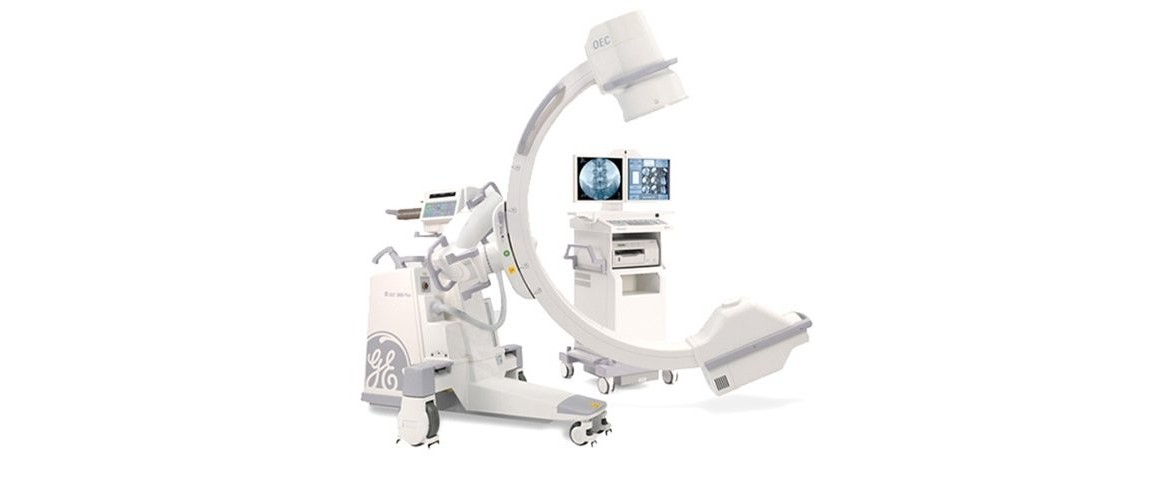
The GE OEC 9800 first started in 1999, and the upgraded version, the GE OEC 9800 plus, introduced in 2002. It was finally discontinued in 2006 just before the GE C ARM manufacturing facility in Utah claimed the recall by FDA in 2006. There has been no successor of the GE OEC 9800 since 2006. The GE OEC 9900 and the GE OEC 9800 are quite similar in terms of specification and clinical utilities, so GE simplified its product line-up and decided to have the OEC 9900 series with an II (Image Intensifier) type C ARM and a CMOS flat panel detector type CARM. From this perspective, in 2017, GE cleared the 510(k) for their CMOS Flat Detector, now incorporated into the GE OEC Elite CFD as a premium grade C-ARM system. The GE OEC 9900 was upgraded with a 1k x 1k CCD camera and the traditional II type is still importantly positioned as a key C-ARM product in the GE C-ARM line-up.
In Same Category
- The heart-lung machine and its crucial role in open-heart surgeries
- Modern endoscopy - evolution of technology and the role of used parts
- Mammography - the role of used parts in breast cancer diagnostic medical equipment
- Siemens healthineers C-Arm systems - advancing precision in medical imaging
- Philips Veradius C-arm Advanced Imaging and Sustainable Solutions in Medical Equipment

Comments
Leave your comment